Bradley Ladewig is Paul Wurth Chair in Energy Process Engineering at the University of Luxembourg. His teaching and research interests are in hydrogen technologies, especially in energy applications.
Download my CV .
Experience
Paul Wurth Chair in Energy Process Engineering
Responsibilities include:
- Research in hydrogen materials and technologies
- Teaching in the Bachelor and Masters of Engineering
- Industry engagement especially in pan-European hydrogen projects
Visiting Professor
Group Leader
Senior Lecturer in Chemical Engineering
Associate Professor of Chemical Engineering
Appointed Lecturer in 2009, promoted to Senior Lecturer in 2011 and Associate Professor in 2012
Projects
Luxembourg Hydrogen Valley (LuxHyVal) is a proposal to establish a Hydrogen Valley in Luxembourg, using renewable electricity to produce green hydrogen for several industrial and mobility end-users. The project consortium is comprehensive and includes The University of Luxembourg (as coordinating institution and Prof.
Advanced electrochemical conversion of ammonia to hydrogen as part of a global supply chain in green hydrogen. A collaboration between the University of Luxembourg and IIT Bombay, India.
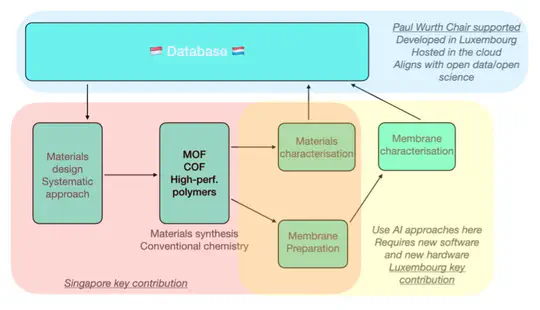
Advanced porous materials for hydrogen separations – smart development and characterisation approaches supported by machine learning - a collaboration between the University of Luxembourg and National University of Singapore
Recent Publications
Quickly discover relevant content by filtering publications.
(2023).
Carbon Capture with Polymeric Membranes.
Reference Module in Earth Systems and Environmental Sciences.
(2023).
Functional role of B-site substitution on the reactivity of CaMFeO3 (M = Cu, Mo, Co) perovskite catalysts in heterogeneous Fenton-like degradation of organic pollutant.
Journal of the Taiwan Institute of Chemical Engineers.
(2022).
Photoresponsive Polymer and Polymer Composite Membranes for Gas Separation.
ACS Applied Polymer Materials.
(2022).
A batch photoreactor for small-scale laboratory research - a technical note.
ChemRxiv.
(2022).
Natural and recycled materials for sustainable membrane modification: recent trends and prospects.
Science of the Total Environmental.
Contact
The Paul Wurth Chair is located at both the Kirchberg and Belval campuses of the University of Luxembourg.
- bradley.ladewig@uni.lu
- 2 Av. de l'Universite, 4365 Esch-sur-Alzette, Luxembourg, sch-sur-Alzette, 4365